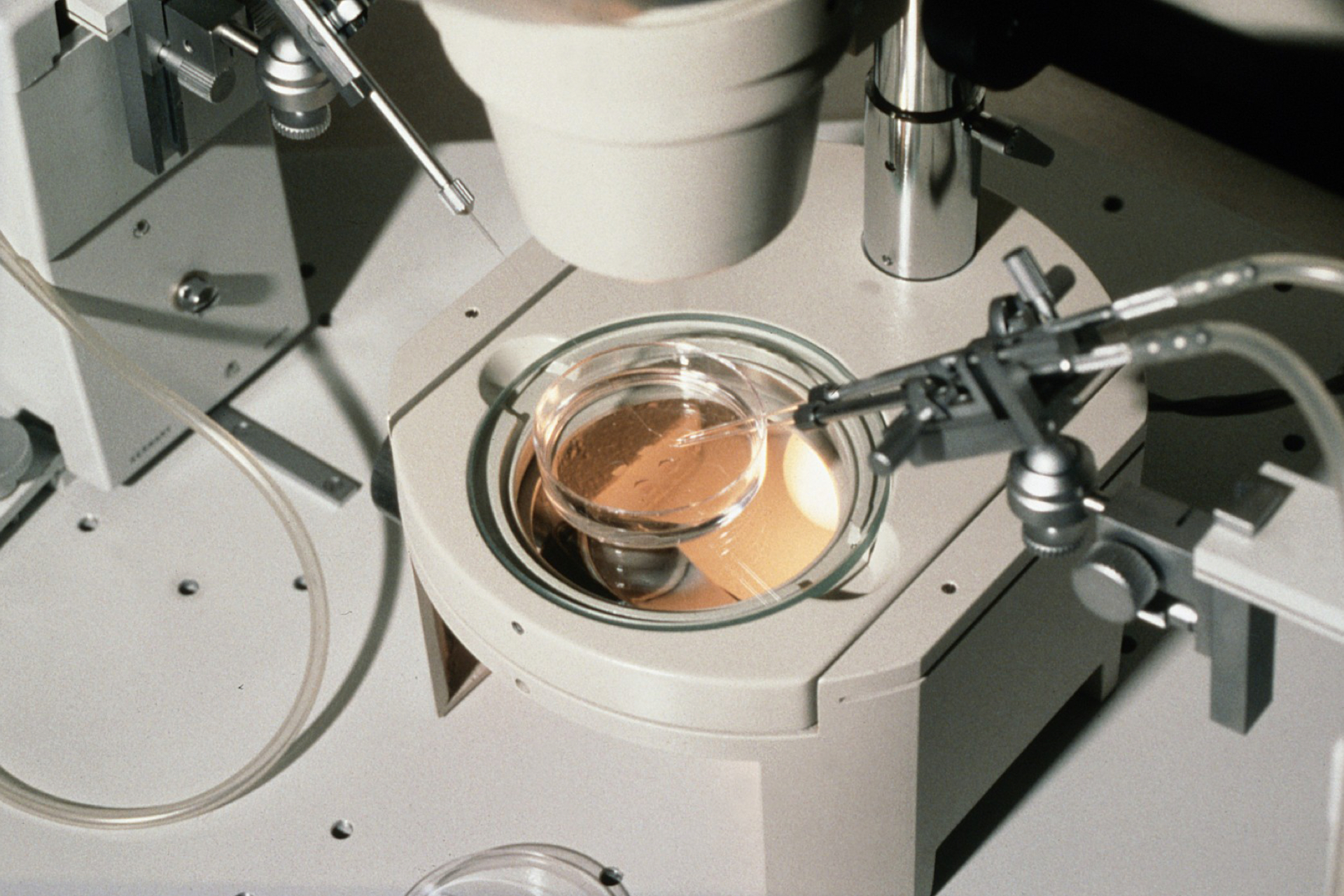
US scientists have carried out the world's first successful ovary tissue transplant in a primate, and have used one of the resulting (1680, eggs)} to produce a healthy IVF baby monkey. Team leader David Lee, of Oregon University, said it was the first time transplanted ovarian tissue had been used to create a healthy infant. He said that the procedure could be used to preserve the fertility of cancer survivors, to treat early menopause, and also 'suggests that ovarian tissue banking in humans may be feasible'. He presented the results at the annual American Society for Reproductive Medicine, being held in San Antonio, Texas this week.
To carry out the procedure, the researchers removed the ovaries from seven rhesus macaque monkeys, and re-implanted slices of tissue into either the kidney, arm or the abdomen. All the monkeys then began producing female hormones, and four also produced eggs, of which two were fertilised using ICSI (intracytoplasmic sperm injection). These embryos were returned to the womb of a surrogate mother, and a healthy female infant was born last year. Although scientists have already successfully carried out ovarian transplants resulting in live births in rats and sheep, this is apparently the first time the procedure has worked in a primate. 'This experiment brings us a little closer to doing it in humans' said ovarian transplant expert Roger Gosden.
Ovary-tissue banking could help female cancer patients who have to undergo radiotherapy, chemotherapy or ovary removal: treatment that can leave them infertile. Ovarian tissue survives freezing and thawing much more readily than eggs, and some women are already freezing such tissue before cancer treatment, in the hope that they can use it in the future. But so far, the technique has not yet succeeded in human trials.
Despite UK media speculation following reports of the US team's success, many scientists are sceptical that the procedure could one day be used by healthy women to store ovarian tissue, to allow them to have a baby in their late 40s or 50s. 'We're a long way from that' Oregon researcher Nancy Klein told the BBC. 'There's a great loss rate of the number of eggs that will survive the freezing process, and so it requires usually the removal of a whole ovary. Women who are already in their mid-30s probably won't have enough eggs to make that a feasible approach' she said.
Sources and References
-
First successful ovary transplant
-
Monkey that offers hope to the childless
-
Fertility first with tissue transplant
-
Ovary transplant gives hope for cancer patients




Leave a Reply
You must be logged in to post a comment.