In the first session of the 2023 PET (Progress Educational Trust) Annual Conference – 'How Much Change Do We Want? Updating Fertility, Embryo and Surrogacy Law' – the audience was treated to a historical tour de force from Professor Peter Braude, emeritus professor of obstetrics and gynaecology at King's College London.
Professor Braude's opening presentation, entitled 'How Did We Get Here? The Story of the Statute', examined how the Human Fertilisation and Embryology (HFE) Act came into being.
Sharing the Human Fertilisation and Embryology Authority (HFEA) website's 'Timeline of the history of IVF in the UK' infographic, Professor Braude explained that starting from the birth of Louise Brown in 1978, it took 12 years for the first legislation to be enacted in 1990. This, with various updates, lasted a further 18 years before the first thoroughgoing revisions were made in 2008. He explained that the law we currently have was not designed to inhibit either research or clinical practice, and is uniquely 'both permissive and restrictive'.
Now, a further 15 years later, we have another review of the Act proposed (see BioNews 1216). But the focus of Professor Braude's talk was just the progress made towards the original 1990 Act – a mere four milestones in the HFEA infographic!
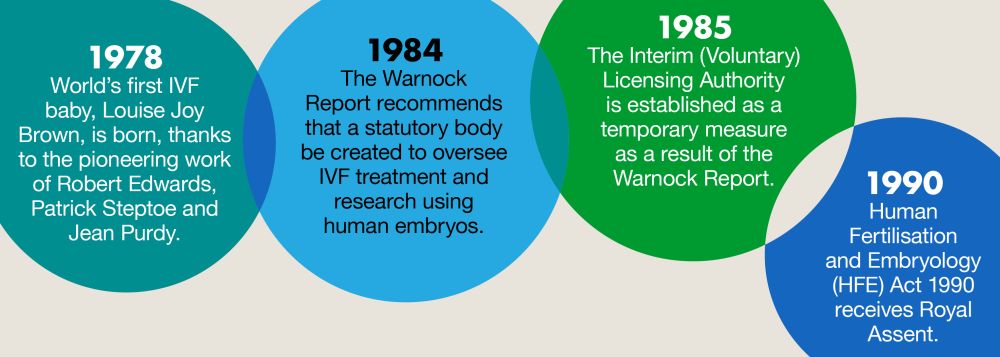
The first milestone showed the birth of Louise Brown in 1978, but, asked Professor Braude, did it really start there? As he explained, the first 'human IVF' was accomplished in 1968, and the paper describing this achievement was published by Professor Sir Robert Edwards in the year of the moon landing: 1969, with the second paper following soon afterwards in 1970. He then described how criticism of IVF preceded Louise Brown's birth by many years and has continued since, as it has never been accepted by some religious groups, and especially not the Catholic Church because of the view that personhood begins at conception (now interpreted as fertilisation). Despite being awarded the Nobel Prize in 2010 (which many lobbied against), Professor Edwards was continually castigated for his work.
Professor Braude went on to show a collage of faces: the dramatis personae involved in the build-up to and passage of the 1990 Act. He pointed out a few, starting with gynaecologist Patrick Steptoe, Professor Edwards and embryologist and nurse Jean Purdy.
Indeed, he went on to say, some may not realise that there were two documented IVF pregnancies that preceded Louise Brown, one in 1973 in Melbourne and another being Steptoe and Edwards' first success in 1976 – providing proof of concept that pregnancy, could result, though at that point, not live birth. Commenting on the reactions to Louise Brown's birth, Professor Braude said the only thing he could think of to compare was Dr He Jiankui's announcement at the 2nd International Summit of Genome editing in 2018 of the birth of two genome-edited babies (see BioNews 977).
And then advances came thick and fast, with other babies following in Australia and the USA. But the UK press and indeed some of the profession remained unimpressed. Even the doctor's trade union, the British Medical Association (BMA), considered experiments on embryos 'unprofessional'. As Professor Braude explained, to 'quell the hysteria and the fear that the science is getting ahead of itself – the Government does what governments do when they don't know what to do: it appointed a commission'.
The Committee of Inquiry into Human Fertilisation and Embryology – a multidisciplinary committee of 15 and a chair – was established under the lead of moral philosopher Baroness Mary Warnock (who would later became the patron of PET). Its remit was to consider the scientific and medical advances and the social, ethical and legal implications of these developments, and then to make policy recommendations. As part of this, it had to examine whether embryo research should be allowed and, if so, until what stage.
Of course, while the Committee debated, IVF advances continued. These included embryo freezing (with the first live birth following freezing and thawing of an embryo reported in 1984), and the use of donated eggs (with a live birth following egg donation reported for a woman with premature ovarian failure in 1983). When the Warnock Committee reported in 1984, it made 63 recommendations. Professor Braude highlighted three key ones: that human embryos should be afforded some protection in law; requirements for legislation rather than guidance; and that a statutory body should be established to licence and monitor fertility treatment and embryo research. Professor Braude described the report as 'eloquent on the research and status of the embryo and its protection, but light on patients and their treatments, few as they were at the time'. Importantly, the Report recommended what has come to be known as 'the 14-day rule', which makes it illegal to handle or conduct research on a live human embryo beyond 14 days post-fertilisation (not including time during which the embryo is frozen).
The Report was welcomed by the Medical Research Council (MRC), the Royal Society, the BMA and the Church of England, but was vigorously opposed by pro-life groups, the Catholic Church and many politicians. There were some calls for a moratorium on embryo research. The MRC and the Royal College of Obstetricians and Gynaecologists accepted the need for regulation, and were already in the process of setting up a Voluntary Licensing Authority (VLA), the first meeting of which took place three days 'before the next bombshell'.
Professor Braude's 'bombshell' was the Unborn Child (Protection) Bill 1984. Enoch Powell MP's Private Member's Bill was selected for debate, having been pushed to the top by anti-abortion supporters who withdrew many of their bills to make way for his. It proposed that possession of a human embryo other than for transfer to a patient would be a criminal offence, no embryo was to be used for research, and the fertilisation of a human egg in vitro would be allowed only with the authority of the Secretary of State who would oversee all IVF treatment.
On its second reading, in February 1985, the Unborn Child (Protection) Bill passed by 238 votes to 66. However, there was much concern that this did not reflect public opinion (as well as the impact it would have on future fertility treatment). By this time, too, the MRC had funded two embryo research programmes in the UK, which would become illegal if the Bill became law. The scientists concerned – one of whom was Professor Braude – wrote to the MRC highlighting the issue. He explained that the MRC response was positive, allowing him to convene a group of scientists and clinicians to provide information and advice to MPs who were about to deal with amendments in committee for the Powell Bill (many of whom are familiar names!). That group (and others) wrote briefs for MPs, sitting in Parliamentary corridors though the night dealing with amendments to soften or overturn the Bill. 'It was an exhilarating and nervous time', said Professor Braude. At the final report stage, debate was 'furious and angry', and long speeches were made with the intention of not allowing the Bill to go to a vote. Consequently, the bill ran out of time.
Meanwhile, explained Professor Braude, 'while Britain was procrastinating', IVF continued across Europe, USA, Japan and Australia, with about 1000 babies already born. The MRC lobby group became a separate campaign group supporting embryo research in human reproduction, recognising the continuing need for a knowledgeable and committed group to advise MPs on the science and medicine of assisted reproduction and embryology: this was the original Progress Campaign for Research into Human Reproduction (the precursor to PET). Patient treatment and welfare, including issues such as donor insemination, was also supported by another Professional Advisory Group, arising from members of the professional societies. The VLA continued to operate, inspecting and issuing licences.
In December 1986, the UK Government – needing to be seen to be doing something – issued a consultation on the establishment of a Statutory Licensing Authority (SLA), with one option being that this be established according to the Warnock Committee's recommendations. Most responses (70 percent) supported this. A later 1987 White Paper set out the proposed remit, function, composition and licensing power of the SLA. In December 1988, the VLA became the 'Interim Licensing Authority' (ILA), and a non-binding debate on the framework for legislation was allowed in both houses. This too was extremely polarised, and the anti-abortion lobby seized the opportunity to try to present a variety of amendments. Concern about research on embryos and IVF was 'stoked by the continual stirring by the press and others of the fears of genetic screening and manipulation'. But then, something happened: as Professor Braude explained, 'a key factor in the reduction of antipathy to research and IVF was the fortuitous announcement by the Hammersmith group in achieving births after PGD coincident with the debates', adding that 'the PGD case was overwhelming and hard to vote against and clearly needed research for it to continue'.
So, what happened? The Human Fertilisation and Embryology Act was given Royal Assent in November 1990. The HFEA was formed in 1991. The Act gave some protection to the embryo in vitro, making it 'a criminal offence to create or use an embryo in vitro without a licence', while prioritising the concept of the 'welfare of the child'. However, the Act contained little about care of patients and was predicated on the idea that the majority of IVF would be state funded by the NHS. These are ideas which have fundamentally changed in practice, and some of which were tackled in the subsequent session of the PET Annual Conference, 'Where Do We Go Next? The Case for Change' (see BioNews 1219).
PET would like to thank the sponsors of this session (the Edwards and Steptoe Research Trust Fund) and the other sponsors of its conference (the Anne McLaren Memorial Trust Fund, ESHRE, Hertility, Born Donor Bank, Carrot Fertility, Ferring Pharmaceuticals, Merck, Theramex, the Association of Reproductive and Clinical Scientists, and the Institute of Medical Ethics.).
Register now for PET's next free-to-attend event, Fertility Treatment for Single People: Who Should Pay?, taking place in Edinburgh on Wednesday 10 January 2024.






Leave a Reply
You must be logged in to post a comment.