The recent news that the first baby born to a mother who had her ovarian tissue frozen and then regrafted, was welcome (see BioNews 1168). However, it is important to understand that while this was not the first time the procedure had been performed, there are various legal and financial barriers to patients accessing these services in the UK.
I had in fact provided care to MAM, a patient who went on to have three children following IVF, after having ovarian tissue regrafted in 2016. MAM was born with the life-limiting condition, B thalassaemia major, the most severe form of the condition. She had a curative bone marrow transplant from her carrier brother at the age of eight in 2001.
MAM's mother had read about experimental work regarding ovarian tissue cryopreservation, and so before the routine conditioning chemotherapy pre-transplant, she travelled from Great Ormond Street Hospital, London to Leeds with her daughter for laparoscopic removal of ovarian tissue. The right ovary was removed, and eight pieces prepared and cryopreserved according to the clinically proven protocol developed by Professor Roger Gosden, who is a leading world expert in this field and was based at the University of Leeds at that time.
This young girls' life was normalised after the successful bone marrow transplant, and she went through puberty, experiencing her first period at age 12. Periods were erratic from the start, and she was diagnosed with ovarian failure, hypothyroidism and osteopaenia at 13 years of age. This was as expected following treatment, before her bone marrow transplant, with sterilising doses of the drugs busulphan and cyclophosphamide (causing sterility in over 99 percent of cases).
MAM presented to me at 21 years of age. She was still having erratic periods, was not on hormone replacement therapy (HRT), and was married to a healthy 26-year-old who was not a thalassaemia genetic carrier. The couple wanted to start a family and the only option, given that the couple are muslim (and therefore prohibited to use donor gametes), was an ovarian tissue autotransplant.
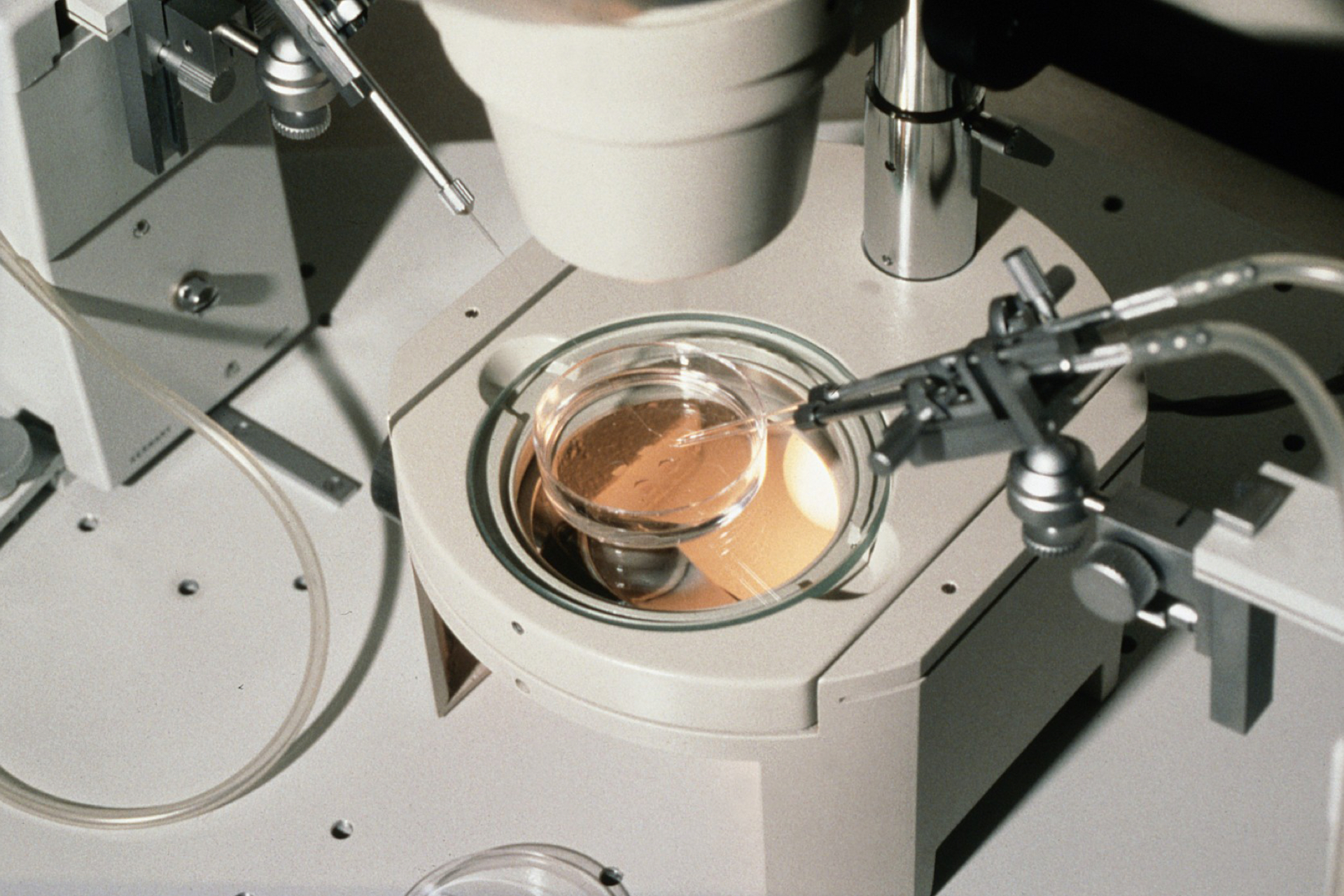
No Human Tissue Authority (HTA)-registered clinic was willing to arrange a transplant for my patient in the UK. Belgium also refused the tissue. Eventually, the experienced team working with Professor Claus Anderson at the University of Copenhagen, Denmark, agreed to help with the surgery, and so the tissue was shipped to Denmark and we performed the very simple surgery to replace four fragments of tissue in August 2015. Three strips were sutured to the remaining denuded ovary cortex, and one was placed within the broad ligament on the side with the absent ovary.
Hormonal activity and follicular growth were evident in the ovary three months later. The fragments placed in the ovary were more active than the one placed in the broad ligament. Ovulation induction at that point produced regular ovulation and periods, but we proceeded with an IVF cycle rather than wait for conception to happen naturally as the couple were still resident in London and keen to go home to Dubai.
Eight eggs were collected, three fertilised and all electively cryopreserved on day two of development. Two embryos were transferred the following month, resulting in a singleton uncomplicated pregnancy and the birth by caesarean section of a healthy baby boy at the Portland Hospital in London in December 2016.
A regular menstrual cycle re-established after stopping breastfeeding and my patient returned to the UK for further fertility treatment in 2019. Laparoscopic adhesiolysis and bilateral salpingostomy was performed to repair bilateral tubal blockage. Two further IVF cycles were performed over the next four months resulting in the banking of three day-five blastocysts.
Two blastocysts were transferred the following month and a healthy baby girl born at home in Dubai nine months later. An unplanned spontaneous pregnancy then followed just after stopping breastfeeding, and another healthy boy completed this couples' family in July 2022.
There are over 200 children born worldwide now following ovarian tissue transplantation. It is the only way to preserve fertility for pre-pubertal girls who need sterilising chemotherapy for either cancer treatment or for conditioning before bone marrow transplant for haematological conditions. It has now been accepted into clinical practice and should be offered to all girls who are at high risk of premature ovarian failure, after lifesaving medical treatment, throughout the UK.
This case highlights several important unique issues:
- Tissue that has been frozen for over 13 years does not deteriorate. The tissue used for this transplant is the longest frozen that has been recorded worldwide.
- Pre-pubertal ovarian tissue adapts immediately and is hormonally responsive when transplanted back into an adult.
- Pre-pubertal grafts can remain active for a prolonged time. Seven years of functioning is exceptional for an ovarian tissue graft, but it makes sense that tissue from younger children should last longer simply due to the increased density of young egg cells.
Restrictions on ovarian tissue transplants
The HTA introduced legislation regarding the conditions of procurement and storage of both ovarian and testicular tissue in 2007. The Human Fertilisation and Embryology Authority previously covered mature gametes only, and so any tissue stored before 2007 is outside any jurisdiction in the UK. There are hundreds of such samples still stored in good faith, predominantly for pre-pubertal and adult cancer patients, in research and IVF laboratories in the UK.
Now there are a handful of centres in the UK which currently offer ovarian tissue storage for selected children and adults under 35 years. There is no NHS funding provision for surgery to collect the tissue, storage, transplant, and any subsequent fertility treatment the patient might need, including IVF. The National Institute for Health and Care Excellence and the HTA are currently in consultation about this issue, and we hope that will bring hope for all affected children with a new diagnosis and all those with tissue stored after 2007.
Unfortunately, it does not address the issue of what to do with the tissue frozen in hope for many young women and children in the UK before 2007. There is no English registry for this tissue and all centres are reluctant to use this tissue, even though it is outside government jurisdiction, although I am hoping to encourage a structure for change.
Several women have approached me about transplanting their frozen tissue since the media coverage of my patient's story in 2016. Denmark has now closed their doors to tissue from the UK, so I am having to go further afield for the simple transplant surgery which the patients need to self-fund completely. It seems stupid when the patients will come back into the UK for their fertility treatment and management afterwards. A better solution would be a select multi-disciplinary committee that can help decide whether the tissue is suitable for use or not, and then it should be transplanted back, and funded by the NHS, in an HTA-recognised facility in the UK.




Leave a Reply
You must be logged in to post a comment.